
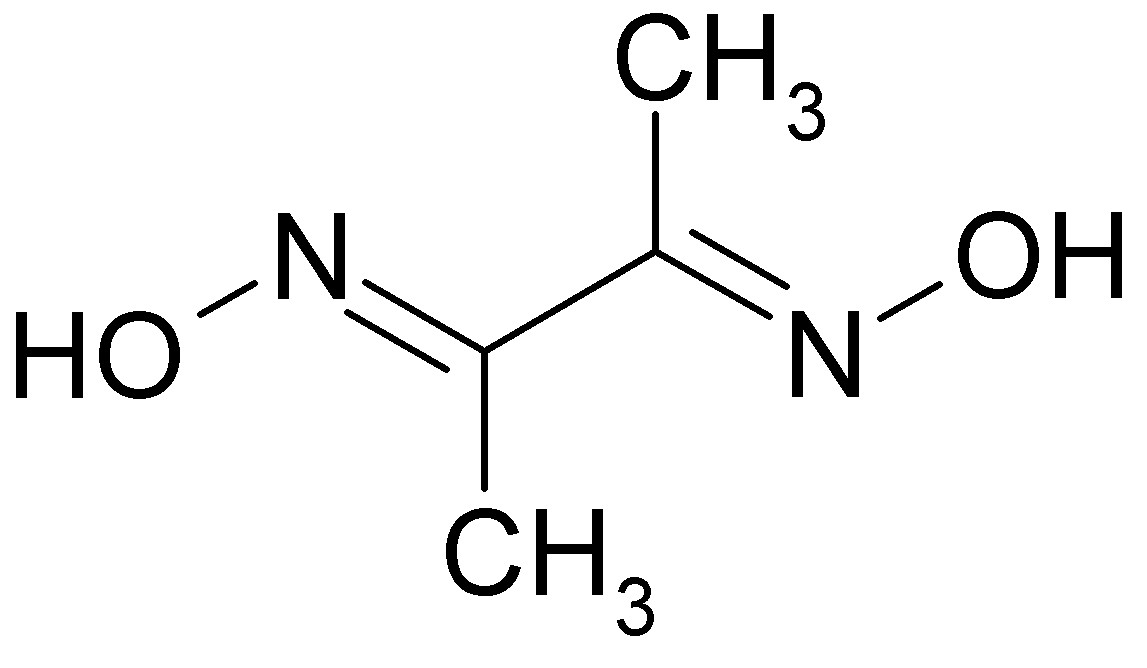
Visit ChemicalBook To find more NICKEL DIMETHYLGLYOXIME(13478-93-8) information like chemical properties,Structure,melting point,boiling point,density,molecular formula,molecular weight, physical properties,toxicity information,customs codes. You can also browse global suppliers,vendor,prices,Price,manufacturers of NICKEL DIMETHYLGLYOXIME(13478-93-8).
Article Views are the COUNTER-compliant sum of full text article downloads since November 2008 (both PDF and HTML) across all institutions and individuals. These metrics are regularly updated to reflect usage leading up to the last few days.Citations are the number of other articles citing this article, calculated by Crossref and updated daily.The Altmetric Attention Score is a quantitative measure of the attention that a research article has received online. Clicking on the donut icon will load a page at altmetric.com with additional details about the score and the social media presence for the given article.
Find more information on. Structural changes in nickel dimethylglyoxime (Ni(dmg) 2) were followed by single-crystal X-ray diffraction in a diamond-anvil cell (DAC) at pressures up to 5.1 GPa, that is, in the pressure range through the major color change point (2 GPa), but before the phase transition at 7.4 GPa. Significant average compression (∼4%/GPa) was observed, with anisotropic, but continuous and monotonic lattice strain. The maximum compression was observed for the direction perpendicular to planar layers of Ni(dmg) 2 and thus corresponds to decreasing the shortest contacts between nickel cations. Compression within the layers was not so pronounced as the compression between the layers. The structure and dynamics of the short O–HO hydrogen bond connecting the adjacent dimethylglyoxime ligands were investigated by periodic DFT calculations and showed evidence of a flat, asymmetric single-well proton potential facilitating large-amplitude proton oscillations. The proton motion appears to be coupled to the dynamics of the adjacent methyl groups, resulting in the increased asymmetry of the hydrogen bond at higher pressures.
